High adsorption capacity of Rhodamine B dye on bio-activated carbon derived from sugarcane bagasse
Department of Chemistry and Environment, Joint Vietnam-Russia Tropical Science and Technology Research Center
63 Nguyen Van Huyen stress, Nghia Do ward, Hanoi, Vietnam
Phone: +84 975482730; Email: hoaiphuong.vrtc@gmail.com
Main Article Content
Abstract
This paper details the synthesis, characterisation, and adsorption efficacy of bio-activated carbon (BAC) generated from sugarcane bagasse for the effective elimination of Rhodamine B (RhB) dye from aqueous solutions. Sugarcane bagasse, an accessible agro-industrial byproduct, was chemically activated with HCl and subsequently carbonized to yield a porous carbon material rich in surface functionalities. Structural and surface analyses validated the formation of a hierarchical mesoporous framework, as demonstrated by SEM, FTIR, XRD, EDX, and nitrogen adsorption–desorption tests, including predominant pore sizes within the 2–5 nm range and intact oxygenated groups conducive to dye adsorption. Batch adsorption tests exhibited fast elimination of RhB, reaching equilibrium in 20 minutes and sustaining removal efficiencies over 95% across a broad concentration spectrum. Kinetic modeling indicated that the pseudo-second-order model most accurately represented the adsorption process, emphasizing chemisorption as the primary mechanism, with heterogeneous surface interactions and pore diffusion effects. Isotherm analysis revealed that the Freundlich model exhibited the optimal fit (R² = 0.9950), indicating multilayer adsorption on energetically heterogeneous sites, with a maximum adsorption capacity (qmax) of 30.36 mg g⁻¹ as determined by the Langmuir model. Further fittings to the Temkin, Elovich, Henry, and Dubinin–Radushkevich models corroborated a synthesis of physical and chemical adsorption mechanisms. The findings validate that sugarcane-bagasse-derived BAC is an economical, sustainable, and efficient adsorbent, presenting considerable promise for practical wastewater treatment applications while facilitating the valorization of agricultural residues into valuable environmental remediation materials.
Keywords
Bio-activated carbon, sugarcane bagasse, rhodamine B removal, adsorption isotherms and kinetics, waste valorization
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Highlights:
Bio-activated carbon derived from sugarcane bagasse achieves >95% Rhodamine B removal within 10–20 minutes, demonstrating fast adsorption kinetics and high affinity.
Adsorption follows a pseudo-second-order kinetic model and fits best with the Freundlich isotherm, indicating chemisorption on heterogeneous surfaces with multilayer formation.
The adsorbent exhibits a high qₘₐₓ of 30.36 mg g⁻¹, with a mesoporous structure and oxygen-rich functional groups, promoting sustainable valorization of agro-industrial sugarcane waste.
1. INTRODUCTION
The textile industry is one of the biggest users of the dyes and accounts for 70% of its market generates approximately one trillion dollars, accounts for 7% of total global exports, and employs approximately 35 M people worldwide [1, 2]. The textile industries are associated with discharge huge quantities of the dye wastewater which are resulted from the washing textiles, coloured or the printed fabrics. The reports indicated that 40,000 industrial synthetic dyes are actively generating 450,000 tons (t) of the dyes annually [3]. One of the most toxic dyes in textile wastewater is Rhodamine B (RhB) which has an extensive value in the textile industry as textile colourant because of its high stability and non-biodegradable [4]. RhB is a synthetic cationic xanthene dye extensively utilized in the textile, printing, and cosmetic sectors [5]. Nonetheless, its discharge into aquatic environments presents significant ecological hazards, including persistence in aquatic ecosystems, phototoxicity, and possible carcinogenic and neurotoxic consequences [4, 6]. Traditional treatment methods, such as coagulation, advanced oxidation, and membrane filtration, can entail significant operational expenses, produce detrimental byproducts, and need intricate infrastructure [7-9]. These gaps offer the opportunity for the researchers to find more efficient technologies for improving the quality of textile wastewater treatment before the final discharge into the environment. Adsorption, especially via activated carbon, has proven to be an effective, adaptable, and eco-friendly method for the elimination of dye pollutants from aqueous solutions (e.g., recent extensive reviews of RhB treatment technologies) [10-12]. Activated carbon (AC) is a widely used material for electrochemical, energy, and environmental applications due to its low cost, large surface area typically ranging from 500 to over 3000 m2 g-1, and developed porous structure with a pore system comprising micropores (< 2 nm), mesopores (2–50 nm), and macropores (> 50 nm), which helps increase its adsorption capacity and chemical reactivity on the surface. However, conventional activated carbon production using coal or petroleum-derived feedstocks is resource-intensive and less sustainable [13-15].
Amid the worsening global environmental pollution and the ongoing energy crisis, the development of sustainable and environmentally benign materials has become a critical research priority. In this context, bio-based activated carbon (Bio-AC), derived from renewable biomass resources such as bark, rice husk, corn cobs, coffee husks, and sugarcane bagasse, has gained considerable attention as an emerging generation of high-performance adsorbents. Owing to its wide availability, low production cost, and tunable physicochemical properties, Bio-AC offers a compelling alternative to conventional activated carbons for environmental remediation applications. AC made from agricultural wastes is considered more efficient due to the high efficiency of removing dyes effortlessness and no hazardous by-products. Conversely, sugarcane bagasse, a byproduct of the sugar and bioethanol industries, provides a sustainable, economical, and plentiful biomass source, containing high levels of cellulose (45–55%), hemicellulose (20-25%), and lignin (18-24%) [16-18]. Bagasse is an ideal low-cost raw material for making activated carbon. Numerous studies have shown the effective transformation of sugarcane bagasse into activated carbon with comparable porosity and adsorption efficacy via chemical or physical activation techniques [19-21].
While activated carbon from sugarcane bagasse has demonstrated effectiveness in adsorbing dyes such as Rhodamine B and heavy metals, research specifically focused on the removal of RhB using bio-activated carbon from the green-skin fraction of sugarcane bagasse is scarce [22-24]. This work contributes significant new insights into the unexploited potential of sugarcane green-skin waste, advances a sustainable route to high-performance activated carbon, and establishes a robust adsorption mechanism for RhB removal. These contributions highlight the feasibility of converting underutilized agro-industrial residues into functional materials for wastewater remediation, supporting both environmental sustainability and circular resource utilization.
2. EXPERIMENTAL
2.1. Materials and reagents
Rhodamine B (RhB, ≥ 99%), potassium hydroxide (KOH, pellets), hydrochloric acid (HCl, 37%), sodium hydroxide (NaOH, pellets), sodium chloride (NaCl, > 99.5%), and ethanol (C2H5OH, ≥ 99.5%) were of analytical grade and utilized as received. Pure water was utilized consistently. Fresh sugarcane bagasse (SCB) was procured from a local juice press, meticulously cleaned with tap water followed by deionized water to eliminate residual sugars and particulates, and subsequently air-dried.
2.2. Preparation of bio-activated carbon (BAC)
The process of preparing BAC is shown in (Figure 1). The cleaned sugarcane bagasse (SCB) was oven-dried at 105°C for 24 h, milled to ≤ 2 mm, and stored in airtight containers with desiccant. For each batch, 20 g SCB was placed in a quartz boat and heated under nitrogen (100 mL min⁻¹), ramped to 450°C at 10°C min⁻¹, held for 2 h, then cooled. The precursor was impregnated with 10 wt% HCl (HCl:SCB = 1:1, w/w), soaked 24 h at room temperature with intermittent stirring, filtered, rinsed with distilled water to neutral pH, and dried at 105oC overnight. Impregnated samples were carbonized at 500°C for 60 min in nitrogen (10°C min⁻¹). The product AC was ground to < 200 µm, stored in desiccant vials, and the biochar yield was determined.
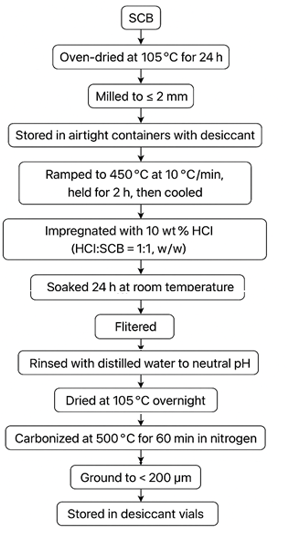
Figure 1: Flowchart showing the process of preparing BAC
2.3. Characterizations
Morphology was analyzed using scanning electron microscopy (SEM). Crystallinity was assessed by X-ray diffraction (XRD) using Cu Kα radiation at 40 kV and 30 mA, over a 2θ range of 10–70°, the phase of the material was closely analyzed at a rate of 5°/min. FTIR infrared spectroscopy was employed to investigate the material's bonds and functional groups in the 400–4000 cm-1 wavenumber region. The elemental composition was ascertained using EDX analysis. The textural qualities were assessed using N₂ adsorption-desorption, measuring the BET surface area, pore volume at P/P₀ = 0.99, the porosity of the materials was comprehensively examined at 77 K, and pore size distribution (BJH) following degassing at 150°C for 3 hours.
2.4. Batch adsorption experiments
A 1000 mg L⁻¹ RhB stock solution was produced in water and stored in the dark at refrigeration temperatures for no more than one week. Daily preparations of working solutions (5–50 mg L⁻¹) were conducted. UV-Vis spectrophotometry at λmax = 552 nm (confirmed by scanning from 400 to 700 nm) was employed to measure RhB. Calibration curves (minimum of 6 points) exhibited linearity with R² values of at least 0.999; blanks (water) and quality control checks (mid-range standards) were conducted every 10 samples. Supernatants were subjected to filtration using 0.22 µm PTFE before to measurement. Unless specified: V = 50 mL, adsorbent dose = 0.5–2.0 g L⁻¹ (typically 1.0 g L⁻¹), agitation = 150 rpm in 100 mL Pyrex conical flasks. After contact, suspensions were filtered and analyzed by UV-Vis. Adsorption capacity qt and removal efficiency H were calculated by:

qt (mg g⁻¹) is adsorption capacity at time t; Co and Ct (mg L⁻¹) are initial and time-t RhB concentrations; V (L) is solution volume; m (mg) is adsorbent mass; H (%) removal efficiency.
Kinetics study: The initial concentration is C₀ = 10 mg L⁻¹, and the dosage is 1.0 g L⁻¹. Aliquots were collected at many time intervals from 0 to 120 minutes, with concentrated early sampling at 10, 20, 30, 40, 50, 60, 90, and 120 minutes. The data will be analyzed using the pseudo-first-order (PFO), pseudo-second-order (PSO), Bangham, and Elovich models. The goodness of fit will be assessed using R² values, and the multilinear regions for diffusion will be analyzed.
Isotherm study: Equilibrate solutions at a concentration of 1.0 g L⁻¹ for 24 hours, as confirmed by kinetics, utilizing beginning concentrations (C₀) between 5 and 25 mg L⁻¹. Apply non-linear regression to fit the data to the subsequent isotherm models: Langmuir, Freundlich, Temkin, Elovich, Henry, and Dubinin–Radushkevich. Provide the parameters qmax, KL, KF, and 1/n, together with their respective 90% confidence intervals.
3. RESULTS
3.1. Characterization of bio-activated carbon

Figure 2. SEM images of bio-activated carbon from sugarcane bagasse
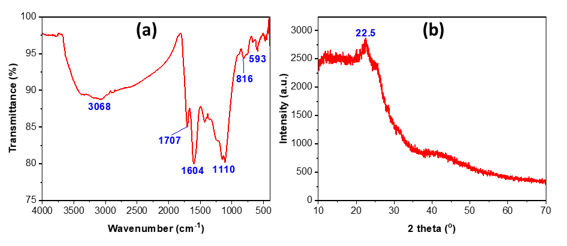
Figure 3. FTIR spectra (a) and XRD pattern (b) of bio-activated carbon from sugarcane bagasse.
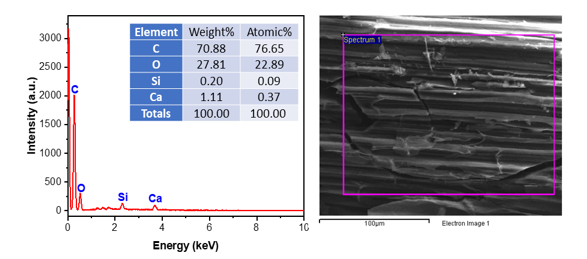
Figure 4. EDX spectra of bio-activated carbon from sugarcane bagasse
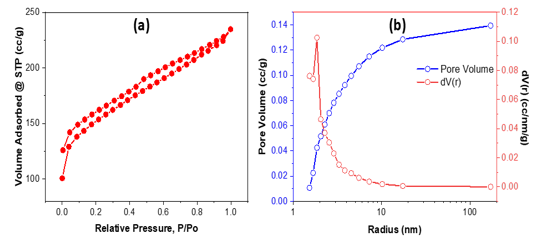
Figure 5. N2 isotherm (a) and pore size distribution (b) of bio-activated carbon from sugarcane bagasse
3.2. RhB adsorption study
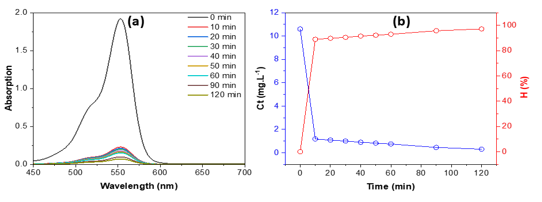
Figure 6. UV-Vis spectra (a), concentration, and removal efficiency (b) of RhB on bio-activated carbon from sugarcane bagasse vs. time
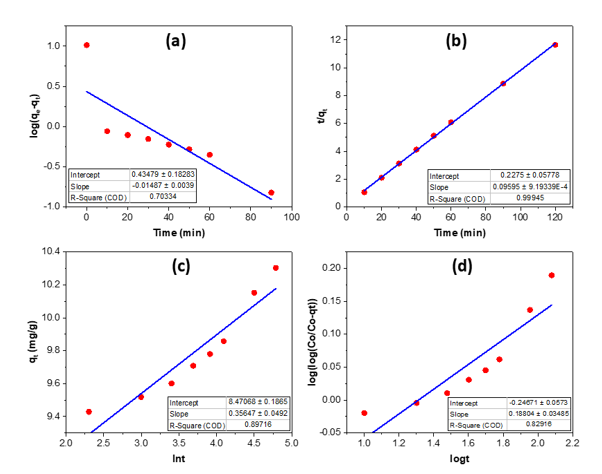
Figure 7. Kinetics of RhB adsorption on bio-activated carbon from sugarcane bagasse: pseudo-first-order (a), pseudo-second-order (b), Elovich (c), and Bangham (d)
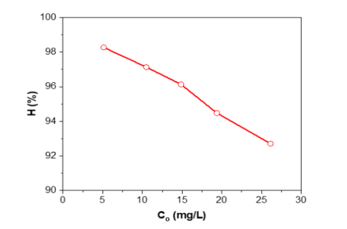
Figure 8. RhB removal efficiency vs. intial concentration by bio-activated carbon from sugarcane bagasse
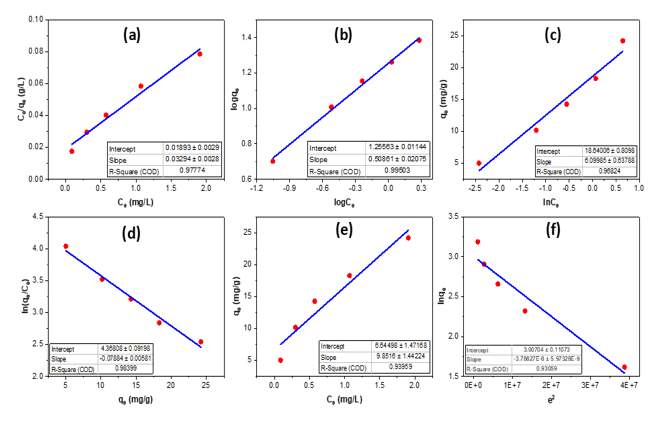
Figure 9. RhB isotherm adsorption on bio-activated carbon from sugarcane bagasse: Langmuir (a), Freudlich (b), Temkin (c), Elovich (d), Henry (e), and Dubinin–Radushkevich (d).
Table 1. Parameters of RhB isotherm adsorption on bio-activated carbon from sugarcane bagasse.
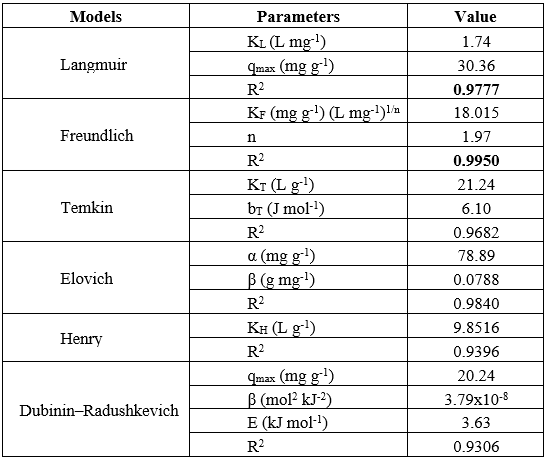
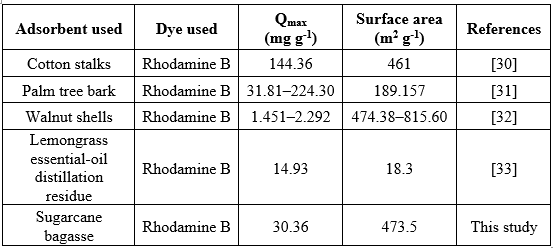
Table 2. Maximum monolayer adsorption capacity and surface area of different agricultural wastes for RhB.
4. DISCUSSION
4.1. Characterization of bio-activated carbon
Figure 2 presents SEM pictures that illustrate the morphological features of bio-activated carbon obtained from sugarcane bagasse. At low magnification (a), the material displays a stratified and uneven architecture with superimposed carbon sheets, signifying the partial retention of the fibrous framework of the predecessor. The layers are loosely organized, resulting in interstitial spaces that can promote adsorptive interactions. At increased magnification (b), the surface is more distinct, revealing elongated, smooth, and aligned carbon structures interspersed with occasional cracks and pores. The activation method improved surface roughness and created porosity, both essential for augmenting surface area and adsorption capacity, rendering the bio-activated carbon appropriate for environmental remediation applications.
Overall, the SEM images confirm that sugarcane-bagasse-derived activated carbon retains a hierarchical porous structure combining macroporous channels with layered microstructures. Such morphology is advantageous for environmental remediation, as it promotes fast adsorption, large accessible surface area, and efficient interaction between pollutants and the carbon framework.
The FTIR spectrum (Figure 3a) of activated carbon synthesized from sugarcane bagasse reveals several characteristic absorption bands, confirming the presence of functional groups typically associated with lignocellulosic-derived carbon materials. The weak band in the range of 3000–3200 cm⁻¹ is attributed to residual O–H stretching vibrations, originating from hydroxyl groups in cellulose, hemicellulose, or surface-adsorbed water. The peak at 1707 cm⁻¹ corresponds to C=O stretching of carbonyl and carboxyl functional groups, indicating partial oxidation during activation. Such oxygen-containing groups are known to enhance the hydrophilicity and adsorption affinity of activated carbon toward polar contaminants. The band at 1604 cm⁻¹ are associated with aromatic C=C stretching, reflecting the development of conjugated carbon structures during pyrolysis. The sharp band at 1110 cm⁻¹ is characteristic of C–O–C stretching in ether or alcohol groups, typically retained from the lignocellulosic matrix. The low-frequency peaks at ~816 cm⁻¹ and ~593 cm⁻¹ correspond to out-of-plane C–H bending and deformation vibrations of aromatic rings, further confirming the formation of an aromatic carbon network [25, 26]. The XRD pattern (Figure 3b) often exhibits two broad diffraction peaks at 2θ = 22.5° (graphitic (002)), indicating low crystallinity and the existence of amorphous carbon [27]. These spectral and structural characteristics align with previously published research on activated carbons derived from sugarcane bagasse. The absence of sharp, well-defined peaks indicates that the carbon framework lacks long-range order. Such structural disorder is typical of activated carbon synthesized from biomass precursors and is advantageous for adsorption applications due to: A high density of defect sites; Increased surface reactivity and a large distribution of micro-and mesopores. The gradual decrease in intensity toward higher angles further supports the amorphous nature of the material.
The EDX spectrum of bio-activated carbon derived from sugarcane bagasse (Figure 4) indicates that carbon and oxygen are the predominant elements, comprising 70.88 wt% (76.65 at%) and 27.81 wt% (22.89 at%), respectively, which aligns with the lignocellulosic nature of the precursor. Trace amounts of calcium (1.11 wt%) and silicon (0.20 wt%) are discovered, presumably stemming from residual inorganic minerals present in bagasse or injected during activation. The elevated carbon content and oxygen functionality signify effective carbonization while maintaining surface heteroatoms, crucial for adsorption and catalytic performance. The elemental compositions closely correspond with previously documented EDX studies of activated carbons generated from sugarcane, which also exhibit a predominance of C and O, along with trace impurities of Si and Ca [26].
The nitrogen adsorption–desorption isotherm (Figure 5a) displays a type IV pattern with an H3 hysteresis loop, indicative of mesoporous materials [28]. The BJH pore size distribution (Figure 5b) indicates a predominant pore radius of around 2–5 nm, with additional mesopores exceeding 50 nm, so affirming a hierarchical pore structure. The overall pore volume of around 0.14 cm3 g-1 signifies well-developed porosity conducive to adsorption. This porous architecture plays a crucial role in improving Rhodamine B adsorption by increasing the number of accessible sites and facilitating rapid transport of dye molecules into the interior of the adsorbent structure. The BET surface area of 473.766 m² g-1, indicating extensive porosity and a high surface area suitable for adsorption applications. These characteristics closely mirror those of previously documented sugarcane-bagasse activated carbons, which also exhibit type IV isotherms and mesoporous distributions conducive to pollutant adsorption.
4.2. RhB adsorption study
The UV-Vis spectra of RhB (Figure 6a) exhibit a gradual decline in the absorption peak at 552 nm as contact time increases, signifying the incremental elimination of dye molecules by the bio-activated carbon sourced from sugarcane bagasse. A substantial decrease in absorbance is noted within the initial 10 minutes, indicating fast adsorption of RhB onto the active sites of the adsorbent. After 20 minutes, only negligible alterations transpire, indicating that equilibrium is almost attained. The associated concentration–time profile (Figure 6b) corroborates this trend, with Ct declining sharply from 10 mg L⁻¹ to practically zero, while the removal efficiency (H) swiftly ascends to over 95% within 10 minutes and subsequently stabilizes near 100%. The results underscore the significant affinity and elevated adsorption capacity of bio-activated carbon for RhB, rendering it an exceptionally effective adsorbent for dye-laden wastewater treatment.
Figure 7 illustrates four kinetic models applied to the adsorption of RhB by bio-activated carbon derived from sugarcane bagasse. The pseudo-first-order plot (a) demonstrates a poor fit (R² = 0.703), suggesting that RhB adsorption does not adhere to a straightforward physisorption-controlled mechanism. Conversely, the pseudo-second-order model (b) demonstrates exceptional linearity (R² = 0.999), indicating that chemisorption on several active sites is the primary rate-controlling mechanism. The Elovich figure (c) demonstrates a satisfactory match (R² = 0.897), indicating adsorption on a heterogeneous surface characterized by an energetically diverse array of sites. The Bangham analysis (d) produces a slope α ≈ 0.188 and an intercept ≈ -0.247 (R² ≈ 0.829); α < 1 indicates that pore (intraparticle) diffusion is a considerable contributor, although it is not the exclusive rate-limiting factor-external film diffusion and surface reaction also exert influence. The adsorption of RhB is optimally characterized by the pseudo-second-order model, incorporating heterogeneous-surface effects and mixed diffusion mechanisms.
Figure 8 depicts the influence of the initial RhB concentration on the removal efficiency (H) of bioactivated carbon obtained from sugarcane bagasse. At low concentrations (5 mg/L), the efficiency is exceptionally high (~98%), indicating a surplus of accessible active sites compared to dye molecules. As the initial concentration escalates from 10 to 25 mg/L, H progressively diminishes to approximately 92%, signifying site saturation and competition among dye molecules for finite adsorption sites. Notwithstanding this reduction, removal efficiency persists over 90%, illustrating the robust adsorption capacity of the bio-activated carbon and its efficacy in effectively removing RhB even at elevated pollutant concentrations.
The adsorption isotherms of RhB on sugarcane-bagasse bio-activated carbon (Figure 9, Table 1) offer significant insights into the adsorption mechanism. The Langmuir model (a) demonstrates a strong match (R² = 0.9777) with qmax = 30.36 mg g⁻¹ and KL = 1.74 L mg⁻¹, indicating monolayer adsorption on a uniform surface. The Freundlich model (b) exhibits the highest correlation (R² = 0.9950), with KF = 18.02 and n = 1.97 (>1), indicating advantageous multilayer adsorption on heterogeneous sites. The Temkin isotherm (c) (R² = 0.9682) signifies a uniform distribution of binding energies and interactions between adsorbate and adsorbent, with KT = 21.24 and bT = 6.10 J mol⁻¹. The Elovich model (d) demonstrates a strong match (R² = 0.9840), indicating chemisorption on heterogeneous surface sites (α = 78.89 mg g⁻¹, β = 0.0788 g mg⁻¹). The Henry model (e) (R² = 0.9396) indicates linear adsorption at low concentrations, whereas the Dubinin–Radushkevich model (f) (R² = 0.9306) reveals qmax = 20.24 mg g⁻¹ and E = 3.63 kJ mol⁻¹, signifying the preponderance of physical adsorption. Adsorption is most accurately characterized by the Freundlich model, which emphasizes heterogeneity and multilayer coverage. The adsorption behavior of Rhodamine B on the prepared activated carbon is governed by a combined physical–surface-controlled mechanism rather than purely chemisorption.
Various agricultural wastes have been investigated as low-cost adsorbents during the previous few decades as shown in Table 2. It can be observed that some materials, such as activated carbon from cotton stalks and palm tree bark, exhibit higher qₘₐₓ values than that obtained in the present study. This variation is mainly attributed to differences in precursor composition, activation method, surface area, and pore structure. Although the maximum adsorption capacity of sugarcane bagasse–derived bio-activated carbon (30.36 mg g⁻¹) is moderate compared with certain reported adsorbents, it demonstrates rapid adsorption kinetics, achieving over 95% dye removal within a short contact time, and maintains high removal efficiency across a wide concentration range. Moreover, the preparation process employs readily available agro-industrial waste and mild activation conditions, contributing to lower production cost and environmental impact. Therefore, rather than outperforming all reported materials in terms of adsorption capacity, sugarcane bagasse-derived bio-activated carbon offers a competitive and sustainable alternative for dye removal, particularly suitable for practical wastewater treatment applications where cost, availability, and treatment efficiency are critical considerations.
5. CONCLUSION
The effective synthesis and application of bio-activated carbon (BAC) derived from sugarcane bagasse for the removal of Rhodamine B (RhB) from aqueous solutions are demonstrated. Comprehensive characterization confirms the formation of a porous carbon structure with abundant surface functional groups and favorable elemental composition, which collectively promote efficient dye adsorption. Batch adsorption experiments indicate rapid uptake, with equilibrium achieved within a short contact time and removal efficiencies exceeding 95% under the investigated conditions. Kinetic analysis shows that the pseudo-second-order model best describes the adsorption process, suggesting surface-controlled adsorption kinetics involving heterogeneous active sites. Isotherm modeling indicates that the Freundlich model provides the best fit, reflecting multilayer adsorption on a heterogeneous surface, while results from other models support the coexistence of physical adsorption mechanisms. Although the adsorption capacity (qmax = 30.36 mg g⁻¹) is moderate compared to some reported materials, the use of a sustainable precursor, a simple preparation route, and rapid adsorption behavior highlight the advantages of sugarcane bagasse-derived BAC as a competitive low-cost adsorbent. Further investigation of pH effects and regeneration performance is necessary to fully evaluate its practical applicability in wastewater treatment.
Acknowledgement: This research was conducted within the framework of the project: “Research on the fabrication of TiO2/TCPP composite materials based on activated carbon from sugarcane bagasse for application in treating some PFOS/PFOA compounds in aquatic environment” - No. MT.Đ1.15/25. Funding was provided by the Joint Vietnam-Russia Tropical Science and Technology Research Center.
Statement on the use of Generative AI: AI tools were used only for language editing and not for generating scientific content. All data, analyses, and interpretations were performed and verified by the authors, who take full responsibility for the manuscript.
Author contributions: Nguyen Thi Thu Hang: conceptualization, methodology, investigation, data curation, formal analysis, writing-original draft; Nguyen Kim Thuy: investigation, experimental work, and data collection; Vu Minh Chau: investigation, characterization analysis (SEM, FTIR, XRD); Cao Phuong Anh: data analysis, visualization, and validation; Han Duy Linh: adsorption experiments, kinetic and isotherm modeling; Dang Minh Quang: data interpretation, statistical analysis; Dao Duy Khanh: material characterization support, technical supervision; Duong Tuan Hung: scientific consultation, methodology refinement; Nguyen Thi Hoai Phuong: conceptualization, supervision, project administration, funding acquisition, writing review & editing.
Conflict of interest statement: We have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.